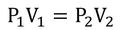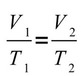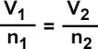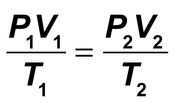Key Concepts
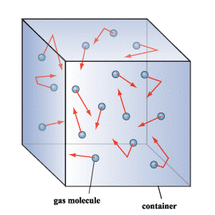
Characteristics of Gases
- A gas always expands to fill its container. Therefore, the volume of a gas equals the volume of the container.
- Gases are highly compressible. When pressure is applied to a gas, its volume decreases.
- Gases are always in constant motion.
- Gases form mixtures with each other regardless of the identities or relative proportions.
Kinetic-Molecular Theory
- Rudolf Clausius
- This theory states that gases are always in constant, random motion and there is no change in energy when molecules collide.
Ideal Gas Law
- PV=nRT
- P- Pressure
- V-Volume
- n- Moles
- R- Gas Constant. It depends on the units used for pressure, temperature, and volume. ( 0.08206 L-atm/mol-K ) or ( 8.314 J/ mol-K )
- T-Temperature
"Ideal Gas Law." YouTube. YouTube, n.d. Web. 29 May 2014.
Partial Pressures
- Dalton's Law- Pt = P1+P2+P3+...
- This equation implies each gas in the mixture behave independently.
"What Is Partial Pressure?" YouTube. YouTube, n.d. Web. 29 May 2014.
STP
- 1 atm = 760 mmHg = 760 torr = 101.325 kPa
The Gas Laws
- Boyle's Law
- Charles's Law
- Avogadro's Law
- Combined Gas Law
"Combined Gas Law." YouTube. YouTube, n.d. Web. 2 May 2014.